| View previous topic :: View next topic |
| Author |
Message |
beallmd
Joined: 10 May 1998
Posts: 1154
|
 Posted: Tue Nov 11, 2014 4:59 pm Post subject: Tahoe Time Posted: Tue Nov 11, 2014 4:59 pm Post subject: Tahoe Time |
 |
|
I'm repeatedly asked about sailing at Tahoe as a Reno regular (Washoe, Lahontan being the usual spots). Well I finally did it a couple weeks ago so I'll report on it; it was certainly a unique and interesting experience. I'll go again when the conditions are right.
First of all the location is at Incline Beach, which is a private beach during the summer for the Hyatt and houses in the community. It is open in the fall some time-not sure when. Very nice little beach and wooded park.
It was cold although not wildly so. Pretty much everyone was in a 4.3. with a hood. A large cloud was coming and finally it rained as the storm arrived. There were a few inches of snow on Mt Rose pass on the way back.
Although it looked really windy it fooled two of us as we under rigged badly. 5.0 to 5.7 would have been correct but it looked like 4.2 sailing with the white water out there.
And that is the first big point. Tahoe is a big deep lake with a straight fetch of about 12 miles so the "chop" was 6 to 8 feet, well overhead in places and breaking in many places. The shore is shallow so breaking waves occur a fair distance out.
Surfers, when I got there, were getting 7 foot faces but that settled down to 5 feet as the day went on. It is surfable and people def surf there.
Second big point; it was a party! It felt like a beach in Maui with 6-10 surfers, 6-8 kiters, 6-8 kayakers and 3 of us windsurfers out at any point; people came and went a lot. The only difference was that it was cold and required heavy suits with hoods etc.
Third big point; this was a really really windy day! Washoe or Lahontan would have been 3.5 0r less sailing. On the way home there were lots of trees and fences blown over (including part of mine). Mt Rose had gusts to about 100!
Summary; Tahoe is big when huge winds come through but not anywhere as windy as elsewhere (I've always heard that). Swell and waves are big, impressive. Sure it's cold but lots of folks take part in it. I'll absolutely go again.
|
|
| Back to top |
|
 |
beallmd
Joined: 10 May 1998
Posts: 1154
|
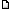 Posted: Tue Nov 11, 2014 5:02 pm Post subject: Posted: Tue Nov 11, 2014 5:02 pm Post subject: |
 |
|
Oh yeah, photo.
| Description: |
|
| Filesize: |
72.26 KB |
| Viewed: |
13902 Time(s) |

|
|
|
| Back to top |
|
 |
rigitrite
Joined: 19 Sep 2007
Posts: 520
Location: Kansas City
|
 Posted: Tue Nov 11, 2014 9:46 pm Post subject: Posted: Tue Nov 11, 2014 9:46 pm Post subject: |
 |
|
what was the density altitude? The problem with alpine lakes is that at altitude, the thinner air makes a real difference in sail performance. You definitely have to rig bigger than normal.
_________________
Kansas City |
|
| Back to top |
|
 |
dsgrntlxmply
Joined: 16 Jun 2010
Posts: 255
|
 Posted: Wed Nov 12, 2014 3:02 pm Post subject: Posted: Wed Nov 12, 2014 3:02 pm Post subject: |
 |
|
A quick try with a density altitude calculator at 1897m, assuming temperature of 12C and a couple of other assumptions, gave a relative density of 0.8, which suggests rigging upwards by a factor of 1.25:
3.5 -> 4.4
4.0 -> 5.0
4.5 -> 5.6
5.0 -> 6.3
5.3 -> 6.6
5.5 -> 6.9
5.7 -> 7.2
6.0 -> 7.5
6.4 -> 8.0
7.3 -> 9.1
|
|
| Back to top |
|
 |
beallmd
Joined: 10 May 1998
Posts: 1154
|
 Posted: Wed Nov 12, 2014 3:28 pm Post subject: Posted: Wed Nov 12, 2014 3:28 pm Post subject: |
 |
|
I know the PGA golfers say that when they play in Denver that the ball travels 10% farther. Roughly at 5,200 feet you need 10% more, I think that's about right. Tahoe is a tad higher at 6,200 feet. I think this is in line with the discussion above. We've always thought you needed another half meter at Washoe (4700 more or less).
BTW with that same storm they said on TV there were 23 foot waves on Lake Michigan.
|
|
| Back to top |
|
 |
gybearoo
Joined: 02 Apr 2005
Posts: 8
|
 Posted: Wed Nov 12, 2014 5:34 pm Post subject: Posted: Wed Nov 12, 2014 5:34 pm Post subject: |
 |
|
| I remember learning in aviation ground school that warm wet air has less lift than cold dry air. Something about water molecules being lighter than air molecules. Plus, the pressure is usually lower on an incoming storm. I rarely sail incoming storm winds (Bay Area), as I tend to be disappointed. I'd rather wait for NW clearing winds which are typically the opposite.
|
|
| Back to top |
|
 |
dsgrntlxmply
Joined: 16 Jun 2010
Posts: 255
|
 Posted: Wed Nov 12, 2014 9:36 pm Post subject: Posted: Wed Nov 12, 2014 9:36 pm Post subject: |
 |
|
Model:
Ideal gas law: PV=nRT. Assume a constant pressure P, volume V, temperature T; R is the gas constant. We then have n moles of gas: the number of gas molecules in the volume being considered.
For density of our test volume, we count the molecules (as n), then consider their molecular masses. Nitrogen and oxygen are diatomic: N2 = 28 amu, O2 = 32 amu. Simplified assumption: 80% N2, 20% O2. The average molecule in dry air is 0.8 * 28 + 0.2 * 32 = 28.8 amu. Next consider H2O: 2*1 + 1*16 = 18 amu. This gives a ratio of 0.625 for water vapor. (Masses and percentage composition are rounded for convenience. Argon is omitted from the simplified model. Ideal gas law is a first-order approximation toward real gas behavior.)
Observation:
Wikipedia article "Water vapor" says dry air is 1.27 g/l and water vapor is 0.804 g/l at STP, or a ratio of 0.633 for water vapor.
The simplified model is close enough to the observation: good enough to explain why.
|
|
| Back to top |
|
 |
|
|
You cannot post new topics in this forum
You cannot reply to topics in this forum
You cannot edit your posts in this forum
You cannot delete your posts in this forum
You cannot vote in polls in this forum
You can attach files in this forum
You can download files in this forum
|
|
|

